Enzymes
|
|
|
2H202 → 2H2O + O2 is a chemical reaction that needs activation energy to start. Think about rolling a boulder up a mountain. You must push the boulder to start the rolling. Activation energy provides the "push" needed to start a chemical reaction.
Catalysts lower the activation energy needed to start chemical reactions. This allows catalysts to speed up the reactions. Proteins that function as biological catalysts are called enzymes. Enzymes can be used in synthesis reactions that build molecules and in digestive reactions that break down molecules. Enzymes therefore play a crucial role in the digestion of food. It is important to note that enzymes are not used up during a reaction and can therefore be used in additional reactions.
Most enzymes end in -ase and the name of the enzymes indicates the reaction it catalyzes. For example, the enzyme sucrase breaks downsucrose and the enzyme DNA polymerase builds DNA.
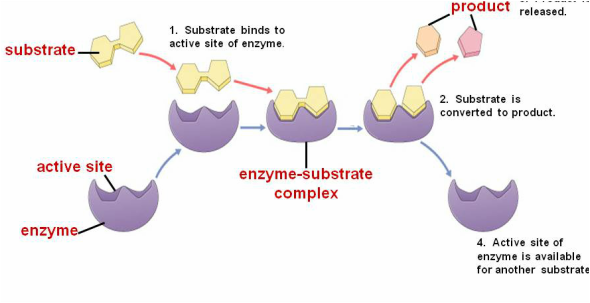
Because of their shapes, enzymes will only catalyze or speed up specific reactions. The lock and key hypothesis offers an explanation of how enzymes work. Substrates or reactants bind to an enzyme at an active site forming an enzyme-substrate complex. The substrate must have a complementary shape to bind to the active site of the enzyme. The substrate and enzyme must fit together like a lock and a key. During the reaction, the substrate undergoes a chemical change and a new substance, the product, is formed. At the end of the reaction, the product is released from the active site. The active site of the enzyme is then free to bond with other substrates to catalyze more reactions.
Remember that the shape of the enzyme determines what substrate it can bind to. A denatured enzyme can not function properly because its shape has been changed. Changes in temperature and pH can change the shape of an enzyme thus causing the enzyme to loose its function.
Catalysts lower the activation energy needed to start chemical reactions. This allows catalysts to speed up the reactions. Proteins that function as biological catalysts are called enzymes. Enzymes can be used in synthesis reactions that build molecules and in digestive reactions that break down molecules. Enzymes therefore play a crucial role in the digestion of food. It is important to note that enzymes are not used up during a reaction and can therefore be used in additional reactions.
Most enzymes end in -ase and the name of the enzymes indicates the reaction it catalyzes. For example, the enzyme sucrase breaks downsucrose and the enzyme DNA polymerase builds DNA.
Because of their shapes, enzymes will only catalyze or speed up specific reactions. The lock and key hypothesis offers an explanation of how enzymes work. Substrates or reactants bind to an enzyme at an active site forming an enzyme-substrate complex. The substrate must have a complementary shape to bind to the active site of the enzyme. The substrate and enzyme must fit together like a lock and a key. During the reaction, the substrate undergoes a chemical change and a new substance, the product, is formed. At the end of the reaction, the product is released from the active site. The active site of the enzyme is then free to bond with other substrates to catalyze more reactions.
Remember that the shape of the enzyme determines what substrate it can bind to. A denatured enzyme can not function properly because its shape has been changed. Changes in temperature and pH can change the shape of an enzyme thus causing the enzyme to loose its function.